The electrochemical conversion of CO2 into carbon-based fuels and valuable feedstocks by renewable electricity is an attractive strategy for addressing CO2 abatement and renewable energy consumption, which is of great significance for achieving the goal of carbon neutralization.
CO is the important component of syngas (a mixture of CO and H2), which can be directly converted into various value-added chemicals via well-developed industrial processes such as Fischer-Tropsch synthesis, methanol synthesis, etc. Therefore, CO2 electroreduction to CO is considered one of the most promising routes to obtain cost-competitive products. However, highly efficient CO2 conversion with high space velocity under mild conditions remains a challenge.
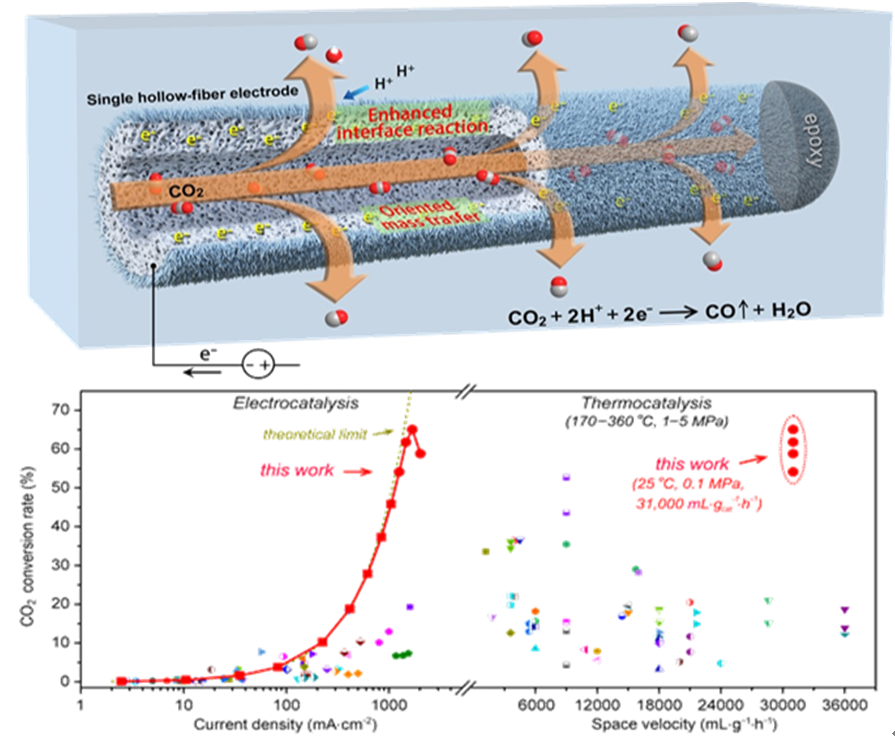
Motivated by such challenge, a research team led by Prof. WEI Wei and CHEN Wei reported a hierarchical micro/nanostructured silver hollow fiber electrode that reduces CO2 to CO with CO2 conversions exceeding 54% at a high space velocity of 31,000 mL · gcat-1 · h-1 under ambient conditions, maintaining stable large current densities (~1.26 A·cm-2) and high CO faradaic efficiencies (~93%). The results were published in the latest Nature Communications.
The reported hollow fiber electrode with hierarchical micro/nanostructures composed of only metallic silver (Ag) for electroreducing CO2 to CO. Such a porous hollow-fiber Ag electrode acting as a CO2 disperser not only enhances three-phase interface reactions but also guides mass transfers during electrolysis. Electrochemical results and time-resolved operando Raman spectra demonstrate that enhanced three-phase interface reactions and oriented mass transfers synergistically boost CO production.
This result provides new opportunities for heightening three-phase interface reactions and mass transfer kinetics simultaneously. In addition, it demonstrates that activated Ag HF can be an ideal industrial electrode with excellent durability, representing an encouraging headway in CO2 electroreduction that may lead to scalable applications.
Source
Shanghai Advanced Research Institute, press release, 2022-06-02.
Supplier
Chinese Academy of Sciences (CAS)
Shanghai Advanced Research Institute (SARI)
Share
Renewable Carbon News – Daily Newsletter
Subscribe to our daily email newsletter – the world's leading newsletter on renewable materials and chemicals














